J.F. Carod1, E. Puget1, P. Pothier1, K. Ambert-Balay2 — Centre Hospitalier Louis Jaillon, Saint-Claude, France,1 Centre National de Référence des Virus Entériques, Dijon, France2
J.F. Carod1, E. Puget1, P. Pothier1, K. Ambert-Balay2 — Centre Hospitalier Louis Jaillon, Saint-Claude, France,1 Centre National de Référence des Virus Entériques, Dijon, France2
Abstract
Enteric vials are known to extend the diagnostic value of stool samples for the detection of enteric pathogens. However, one has yet to be tested for its affect on Immunochromatographic Tests (ICTs) which are widely used to diagnose enteric viruses such as Rotaviruses and Adenoviruses. This study validates the ETM™ vial from Alpha-Tec Systems (Vancouver, WA, USA) for use with the following four ICT assays: RIDA® QUICK Rotavirus/Adenovirus Combi (r-Biopharm), SD BIOLINE ROTA/ADENO® Rapid (Alere, Inc.), VIKIA® Rota-Adeno (bioMérieux Clinical Diagnostics) and ImmunoCard STAT!® Rotavirus (Meridian Bioscience, Inc.). 82 stool samples were collected in clean, sterile vials and were screened with two RT-PCR assays each for the presence of Adenoviruses and Rotaviruses. A portion of the stool was then aliquoted into the ETM vial at a ratio of 1:4, stool to media. Both fresh, untreated samples and samples from the ETM vial were used for the four aforementioned ICTs. Results show 100% concordance between both ICT samples sets and the two RT-PCR assays in that each was able to successfully detect all known positive and negative samples. Thus, it is the conclusion of this study that the ETM shows no inhibitory effects on these ICTs and that sensitivity was not compromised by the sample being diluted into the ETM media.
Background
Rotaviruses and Adenoviruses are responsible for the majority of pediatric diarrhea cases in the world. Immunochromatographic Tests (ICTs) provide a rapid diagnosis of diarrhea due to viral infection. Transport media vials are designed to preserve enteric bacteria present in patient samples, but they have not yet been evaluated for their ability to preserve viruses or their potential effects on correlating ICTs. The ETM vial from Alpha-Tec Systems (Vancouver, WA, USA) was chosen for this study. It is a well-known enteric transport media which consists of a proprietary modified Cary Blair media, a pH indicator to detect the overgrowth of commensal organisms, and a plastic spoon which is attached to the inside lid of the vial. It is designed for collection, transport and preservation of stool specimens. ETM is compatible with a variety of testing methods including agar culture, Enzyme Immuno Assays for Giardia lamblia and Cryptosporidium, and C. difficile toxin detection. Additionally, it is compatible with most specimen processor devices.
Objectives
The objective was to validate ETM media compatibility with four Adenovirus/Rotavirus ICTs by comparing specificity and sensitivity performance against fresh, untreated fecal specimens. The four ICTs to be validated for use with the ETM vial were: RIDA® QUICK Rotavirus/Adenovirus Combi (r-Biopharm), SD BIOLINE ROTA/ADENO® Rapid (Alere, Inc.), VIKIA® Rota-Adeno (bioMérieux Clinical Diagnostics) and ImmunoCard STAT!® Rotavirus (Meridian Bioscience, Inc.).
Materials and Methods
Stool specimens were collected in a clean, sterile vial, and were stored frozen for up to two weeks. Samples were then thawed and an aliquot was taken to test for the presence or absence of Adenoviruses and Rotaviruses with an RT-PCR assay for each analyte 4, 5. The results were recorded and the samples were frozen again for up to one year. Upon a final thawing of the sample, an aliquot was taken and put into the ETM vial yielding a 1:4 dilution of specimen to fixative. Both sets of samples, fresh (untreated) and ETM, were held at room temperature for one hour before testing began with the four ICT assays.
Electron micrographs by F.A. Murphy, University of Texas Medical Branch, Galveston, TX
Results and Conclusion
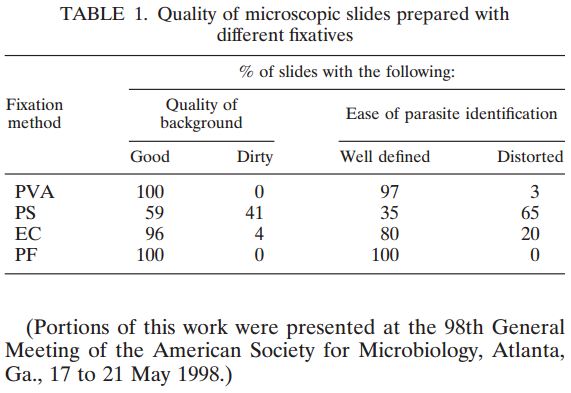
Of the 82 samples collected, 34 were reported as negative, 30 were positive for Rotaviruses, and 18 were positive for Adenovirus. There was 100% concordance of the results between the two RT-PCR assays and both sets of ICT assays. Results indicate that all known samples were correctly identified with all four ICTs in both fresh samples and samples stored in the ETM vial.
| ICT Assay | Sample Type | Detection Rate (Negatives) | Detection Rate (Rotaviruses) | Detection Rate (Adenoviruses) | Concordance |
|---|---|---|---|---|---|
| RIDA® QUICK Rotavirus/Adenovirus Combi | Fresh | 34/34 | 30/30 | 18/18 | 100% |
| ETM | 34/34 | 30/30 | 18/18 | ||
| SD BIOLINE ROTA/ADENO® Rapid | Fresh | 34/34 | 30/30 | 18/18 | 100% |
| ETM | 34/34 | 30/30 | 18/18 | ||
| VIKIA® Rota-Adeno | Fresh | 34/34 | 30/30 | 18/18 | 100% |
| ETM | 34/34 | 30/30 | 18/18 | ||
| ImmunoCard STAT!® Rotavirus | Fresh | 34/34 | 30/30 | 18/18 | 100% |
| ETM | 34/34 | 30/30 | 18/18 |
References
1. Cary, S.G., and E.B. Blair, 1964. “New Transport Medium for Shipment of Clinical Specimens, I. Fecal Specimens.” J Clin Micro 88:96-98.
2. Wang, W.L., et al., 1983. “Evaluation of Transport Media for Campylobacter jejuji in Human Fecal Specimens.” J Clin Micro 18:803-807.
3. Castriciano S., et al., “Validation of Copan Fecal Swab for the detection of rotavirus and adenovirus from fecal specimens using the Coris Rota-Strip and Adeno-Strip rapid antigens tests.” ESCV 2009 Istanbul
4. Allard A., et al., 2001. Rapid Typing of Human Adenoviruses by a General PCR Combined with Restriction Endonuclease Analysis. J Clin Micro 39:498-505
5. Gómara, I., et al., 2002. Molecular Characterization of VP6 Genes of Human Rotavirus Isolates: Correlation of Genogroups with Subgroups and Evidence of Independent Segregation. J Virol 76, 13:6596-6601
ETM™ is a trademark of Alpha-Tec Systems, Inc., Vancouver, WA, USA. www.AlphaTecSystems.com
M-PO-FR3.A